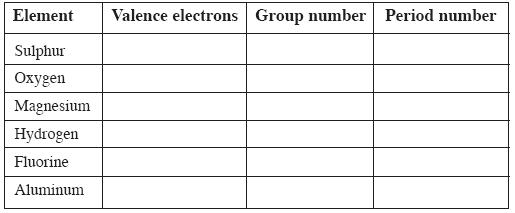
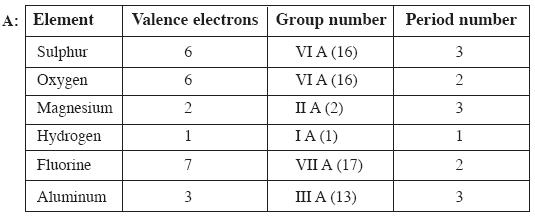
Question: Elements in a group generally possess similar properties, but elements along a period have different properties. How do you explain this statement? (AS – 1) (2 Marks)
Answer: ! Elements in a group have similar electronic configuration. So all the elements in a group possess similar properties. Elements in a period do not have similar electronic configuration. So the elements in a period have different properties and do not have similar properties.
——————————–
Question: s-block and p-block elements except 18th group elements are some times called as ‘Representative elements’ based on their abundant availability in nature. Is it justified? Why? (AS – 1) (4 Marks)
Answer: The statement given is justified. Because Elements in groups 1, 2, 13, 14, 15, 16, 17 of the periodic table are representative elements. These group elements atoms have outer most orbit filled partly or nearly completely with electrons in s and p orbitals.
The 18th group elements atoms are completely filled with electrons in their s and p orbitals and so these do not come under representative elements.
The metals are present in the left group of the representative elements and non – metals are present in the right and semi – metals are present in the middle.
18th group elements are noble gases. The representative elements are all having incomplete outer most orbits. So they are chemically reactive to obtain stable electronic configuration of noble gases. These elements are therefore abundant in nature in the form of compounds.